 In chemistry and medicine, the half-life of a reaction is used to predict the concentration of a chemical over time. “The time it takes for half of the reaction to be completed, i.e., the time in which the reactant concentration is decreased to half of its original value is called the half-life period of the reaction.” The half-life definition of a reaction in simple words can be expressed as What Is the Half-Life Definition of a Reaction? In this module, you learn to perceive the half-life of a reaction and rate equations for zero and first-order reactions. For instance, what controls the rate at which the fuel burns in an auto engine? Which parameters determine how rapidly food gets spoiled? Apart from feasibility and extent, other factors, like the rate of a reaction and the factors that determine the rate, are also crucial for a chemical reaction. How fast is a substance converted into another by a chemical reaction, and is the reaction feasible or not? The extent to which that reaction will proceed is all the questions that decide the course of a chemical reaction. That happens after 10 half-lives.Chemistry is all about changes. The rule is that a sample is safe when its radioactivity falls below detection levels. Knowing about half-lives is useful because it allows you to identify when a radioactive material sample is safe to handle. It will explain why our occupations are becoming more specialised and provide insight into how we may compete more successfully in a highly competitive world. Understanding the notion of a half-life will alter your reading habits and how you spend your time. The half-life of an atom does not explain the precise length of time that each and every atom spends before disintegrating. It is difficult to anticipate the decay of a single radioactive atom. They can then use this information to calculate the age of a drug. They calculate how much of the carbon-14 has been converted. The half-life of carbon-14 can be used by scientists to estimate the age of organic things. The half-life of a radioactive substance is the time it takes for one-half of its atoms to decay. 
The half-life of second-order reactions is affected by both the initial concentration and the rate constant. 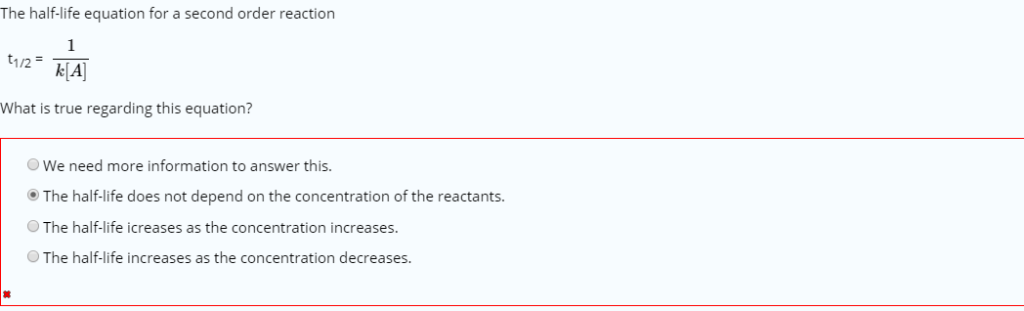
The half-life depends solely on the reaction rate constant k. A zero-order reaction’s half-life is affected by both the starting concentration and the rate constant. To put it another way, increasing the amount of substrate does not increase the pace of the reaction. The rate of a reaction in zero-order kinetics is independent of substrate concentration. We can obtain equations for the half lives for reactions of various orders by substituting the values t=t 1/2 and =1/2 0into the integrated laws Half life of zero order reaction In a first-order reaction, the reactant concentration declines by 1/2 in each sequence of evenly spaced time periods, i.e. Pick any moment in a reaction and one half-life from that point is where the reactant concentration is 1/2. It is worth noting that the half-life is independent of the starting concentration. 
The time it takes for a reactant’s concentration to be lowered to one-half of its starting concentration is referred to as the half-life of a reaction. The reactions are categorised into two classes based on their kinetic properties:Ī) homogeneous reactions, which occur totally in one phase b) heterogeneous reactions, which occur in more than one phase whether the transformation occurs on a catalyst’s surface or the walls of a container. As a result, in chemical kinetics, we can also calculate the rate including a chemical reaction. Such investigations also allow us to comprehend the process through which the response happens. Chemical kinetics is a field of physical chemistry that studies the rate of chemical processes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
